| Record Information |
|---|
| Version | 5.0 |
|---|
| Status | Detected and Quantified |
|---|
| Creation Date | 2006-08-13 03:39:40 UTC |
|---|
| Update Date | 2023-02-21 17:16:48 UTC |
|---|
| HMDB ID | HMDB0003701 |
|---|
| Secondary Accession Numbers | |
|---|
| Metabolite Identification |
|---|
| Common Name | Dimethylbenzimidazole |
|---|
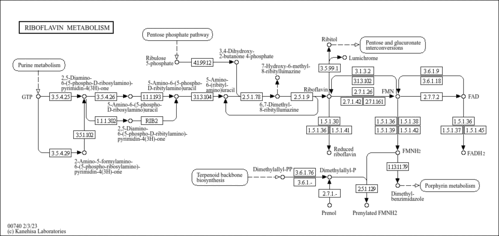
| Description | Dimethylbenzimidazole, also known as dimedazol, belongs to the class of organic compounds known as benzimidazoles. These are organic compounds containing a benzene ring fused to an imidazole ring (five member ring containing a nitrogen atom, 4 carbon atoms, and two double bonds). Dimethylbenzimidazole exists in all living organisms, ranging from bacteria to humans. In humans, dimethylbenzimidazole is involved in the riboflavin metabolism pathway. Dimethylbenzimidazole has been detected, but not quantified in, a few different foods, such as anatidaes (Anatidae), chickens (Gallus gallus), and domestic pigs (Sus scrofa domestica). This could make dimethylbenzimidazole a potential biomarker for the consumption of these foods. Dimethylbenzimidazole is a primary metabolite. Primary metabolites are metabolically or physiologically essential metabolites. They are directly involved in an organism’s growth, development or reproduction. Based on a literature review a significant number of articles have been published on Dimethylbenzimidazole. |
|---|
| Structure | InChI=1S/C9H10N2/c1-6-3-8-9(4-7(6)2)11-5-10-8/h3-5H,1-2H3,(H,10,11) |
|---|
| Synonyms | | Value | Source |
|---|
| Dimedazol | ChEBI | | Dimedazole | ChEBI | | Dimesol | ChEBI | | Dimezol | ChEBI | | 5,6-Dimethylbenzimidazole | Kegg | | 5,6-Dimethyl-1H-benzimidazole | HMDB | | DimezolDimedazol | HMDB | | 5,6-Dimethylbenzimidazole hydrochloride | MeSH, HMDB |
|
|---|
| Chemical Formula | C9H10N2 |
|---|
| Average Molecular Weight | 146.1891 |
|---|
| Monoisotopic Molecular Weight | 146.08439833 |
|---|
| IUPAC Name | 5,6-dimethyl-1H-1,3-benzodiazole |
|---|
| Traditional Name | 5,6-dimethylbenzimidazole |
|---|
| CAS Registry Number | 582-60-5 |
|---|
| SMILES | CC1=CC2=C(C=C1C)N=CN2 |
|---|
| InChI Identifier | InChI=1S/C9H10N2/c1-6-3-8-9(4-7(6)2)11-5-10-8/h3-5H,1-2H3,(H,10,11) |
|---|
| InChI Key | LJUQGASMPRMWIW-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as benzimidazoles. These are organic compounds containing a benzene ring fused to an imidazole ring (five member ring containing a nitrogen atom, 4 carbon atoms, and two double bonds). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzimidazoles |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzimidazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Benzimidazole
- Benzenoid
- Heteroaromatic compound
- Imidazole
- Azole
- Azacycle
- Organic nitrogen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Physiological effect | Not Available |
|---|
| Disposition | |
|---|
| Process | |
|---|
| Role | |
|---|
| Physical Properties |
|---|
| State | Solid |
|---|
| Experimental Molecular Properties | | Property | Value | Reference |
|---|
| Melting Point | 205.5 °C | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | 1.854 | Not Available |
|
|---|
| Experimental Chromatographic Properties | Experimental Collision Cross Sections |
|---|
| Predicted Molecular Properties | |
|---|
| Predicted Chromatographic Properties | Predicted Collision Cross SectionsPredicted Kovats Retention IndicesUnderivatizedDerivatized| Derivative Name / Structure | SMILES | Kovats RI Value | Column Type | Reference |
|---|
| Dimethylbenzimidazole,1TMS,isomer #1 | CC1=CC2=C(C=C1C)N([Si](C)(C)C)C=N2 | 1760.3 | Semi standard non polar | 33892256 | | Dimethylbenzimidazole,1TMS,isomer #1 | CC1=CC2=C(C=C1C)N([Si](C)(C)C)C=N2 | 1689.1 | Standard non polar | 33892256 | | Dimethylbenzimidazole,1TMS,isomer #1 | CC1=CC2=C(C=C1C)N([Si](C)(C)C)C=N2 | 1881.2 | Standard polar | 33892256 | | Dimethylbenzimidazole,1TBDMS,isomer #1 | CC1=CC2=C(C=C1C)N([Si](C)(C)C(C)(C)C)C=N2 | 1999.0 | Semi standard non polar | 33892256 | | Dimethylbenzimidazole,1TBDMS,isomer #1 | CC1=CC2=C(C=C1C)N([Si](C)(C)C(C)(C)C)C=N2 | 1873.3 | Standard non polar | 33892256 | | Dimethylbenzimidazole,1TBDMS,isomer #1 | CC1=CC2=C(C=C1C)N([Si](C)(C)C(C)(C)C)C=N2 | 2026.0 | Standard polar | 33892256 |
|
|---|
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental GC-MS | GC-MS Spectrum - Dimethylbenzimidazole GC-MS (Non-derivatized) | splash10-000t-2900000000-cf01975179c82b56a747 | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - Dimethylbenzimidazole GC-MS (1 TMS) | splash10-0gb9-5790000000-e422b94d0cbe1bd0345e | 2014-06-16 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - Dimethylbenzimidazole GC-MS (Non-derivatized) | splash10-000t-2900000000-cf01975179c82b56a747 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - Dimethylbenzimidazole GC-MS (Non-derivatized) | splash10-0gb9-5790000000-e422b94d0cbe1bd0345e | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - Dimethylbenzimidazole GC-EI-TOF (Non-derivatized) | splash10-000t-1900000000-1cc9a65e6235151e4820 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Experimental GC-MS | GC-MS Spectrum - Dimethylbenzimidazole GC-EI-TOF (Non-derivatized) | splash10-0gb9-2590000000-7f1dc8fd34bdb99d58f6 | 2017-09-12 | HMDB team, MONA, MassBank | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Dimethylbenzimidazole GC-MS (Non-derivatized) - 70eV, Positive | splash10-0002-0900000000-94a00940d804552f77d0 | 2017-09-01 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Dimethylbenzimidazole GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Dimethylbenzimidazole GC-MS (Non-derivatized) - 70eV, Positive | Not Available | 2021-10-12 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole LC-ESI-QQ , positive-QTOF | splash10-0002-0900000000-d879a610883483e63326 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole LC-ESI-QQ , positive-QTOF | splash10-0002-1900000000-7810390c926bc0a2238a | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole LC-ESI-QQ , positive-QTOF | splash10-000t-3900000000-0e438f149c15e3ba2238 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole LC-ESI-QQ , positive-QTOF | splash10-000t-1900000000-286b3359932c2c63dc23 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole LC-ESI-QQ , positive-QTOF | splash10-001i-4900000000-4e9fbfe2a1943cc865e5 | 2017-09-14 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole LC-ESI-QFT , negative-QTOF | splash10-0002-0900000000-4a48df4993767b5c4eff | 2020-07-21 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole LC-ESI-QTOF 35V, negative-QTOF | splash10-0002-0900000000-93fb22bfd73cbaaecda9 | 2020-07-21 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 10V, Positive-QTOF | splash10-0002-0900000000-5f138fc55912e09c04f8 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 35V, Positive-QTOF | splash10-000t-0900000000-0ac7250afc1fd38defe3 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 20V, Positive-QTOF | splash10-0002-0900000000-941f6c90a92fad755dd0 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 40V, Positive-QTOF | splash10-001i-4900000000-9778293bbdebe312995b | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 35V, Negative-QTOF | splash10-0002-0900000000-0581e550bdf15c825ed1 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 40V, Positive-QTOF | splash10-001i-4900000000-f22be5bed413f1b519e8 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 10V, Positive-QTOF | splash10-0002-0900000000-677d27a4db0421a0c22d | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 20V, Positive-QTOF | splash10-0002-0900000000-3c2aee6880c85391a739 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 10V, Negative-QTOF | splash10-0002-0900000000-86d65abcc3c20eeaa9b7 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 20V, Negative-QTOF | splash10-0002-0900000000-25408408c64689b319ee | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Dimethylbenzimidazole 40V, Negative-QTOF | splash10-0006-1900000000-4cefb2993f948a46e8e8 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Dimethylbenzimidazole 10V, Positive-QTOF | splash10-0002-0900000000-92df1f58e19215585f6a | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Dimethylbenzimidazole 20V, Positive-QTOF | splash10-0002-0900000000-a09abdb85c0cd0766165 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Dimethylbenzimidazole 40V, Positive-QTOF | splash10-000t-2900000000-7ad608a4dad05614e995 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Dimethylbenzimidazole 10V, Negative-QTOF | splash10-0002-0900000000-2f5826366923dad35341 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Dimethylbenzimidazole 20V, Negative-QTOF | splash10-0002-0900000000-685b19fc37ae77b7006b | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Dimethylbenzimidazole 40V, Negative-QTOF | splash10-0005-3900000000-1e116130bd866fe7fc71 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Dimethylbenzimidazole 10V, Positive-QTOF | splash10-0002-0900000000-8ffc0ea3ec7f7e0434b8 | 2021-09-22 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-24 | Wishart Lab | View Spectrum |
IR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M-H]-) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+H]+) | 2023-02-03 | FELIX lab | View Spectrum | | Predicted IR Spectrum | IR Ion Spectrum (Predicted IRIS Spectrum, Adduct: [M+Na]+) | 2023-02-03 | FELIX lab | View Spectrum |
|
|---|
| Disease References | | Prostate cancer |
|---|
- Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [PubMed:19212411 ]
|
|
|---|
| General References | - Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM: Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature. 2009 Feb 12;457(7231):910-4. doi: 10.1038/nature07762. [PubMed:19212411 ]
|
|---|