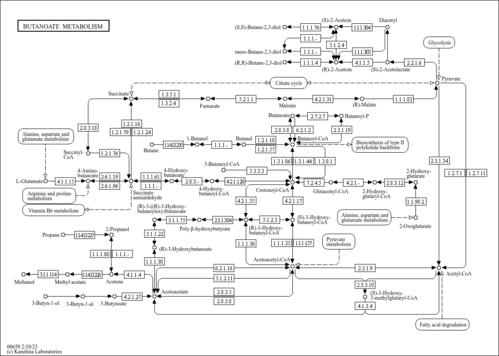
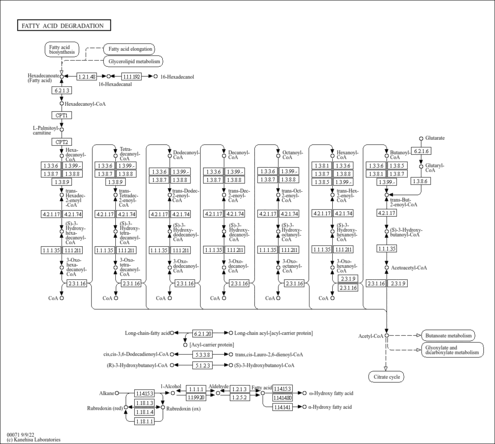
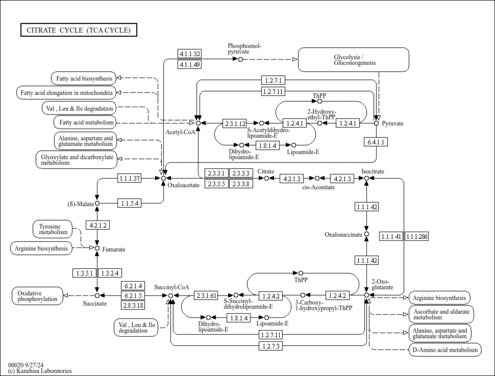
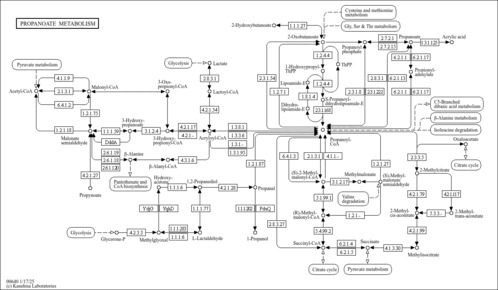
| GC-MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_1) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_2) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_3) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_4) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_5) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_6) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_7) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - Acetyl-CoA GC-MS (TMS_1_8) - 70eV, Positive | Not Available | 2021-10-19 | Wishart Lab | View Spectrum |
MS/MS Spectra| Spectrum Type | Description | Splash Key | Deposition Date | Source | View |
|---|
| Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA LC-ESI-ITFT 35V, positive-QTOF | splash10-0fb9-0035910000-cecfaf54528fc3ef9f00 | 2020-07-21 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA LC-ESI-ITFT 35V, positive-QTOF | splash10-0ufr-0005900000-491971eb7a5c2b327554 | 2020-07-21 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA LC-ESI-ITFT 35V, negative-QTOF | splash10-0a4i-0900000000-1b259612c3897ed851f0 | 2020-07-21 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA LC-ESI-ITFT 35V, negative-QTOF | splash10-08i0-0001901200-8d4f5232c27f2875cf8b | 2020-07-21 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA LC-ESI-ITFT 35V, negative-QTOF | splash10-08i0-0000901200-33f8d3ced8c83336df0c | 2020-07-21 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA 33V, Negative-QTOF | splash10-0a4i-1101800090-5031dc04edf8ba0f3afb | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA 33V, Negative-QTOF | splash10-0a4i-1101900080-6691464da5a030f7d086 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA 33V, Negative-QTOF | splash10-0a4i-1101900080-8a1491c56fc1331c5df1 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Experimental LC-MS/MS | LC-MS/MS Spectrum - Acetyl-CoA 33V, Negative-QTOF | splash10-0a4i-1101900080-9ec3eecc20f19a822002 | 2021-09-20 | HMDB team, MONA | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 10V, Positive-QTOF | splash10-000i-1901000300-57c996f08055dba75dd7 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 20V, Positive-QTOF | splash10-000i-0902000000-dffb00601bfc54014ae4 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 40V, Positive-QTOF | splash10-000i-2901000000-155f0890adf4c76dca85 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 10V, Negative-QTOF | splash10-0arr-6820231930-984ae0f98e0d17e4a7fc | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 20V, Negative-QTOF | splash10-003r-3910100000-87da6b6d742efbc6e74a | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 40V, Negative-QTOF | splash10-057i-5900000000-8701decc3b2311880b97 | 2015-09-15 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 10V, Negative-QTOF | splash10-0a4i-0000000090-fd17a06b039262f96463 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 20V, Negative-QTOF | splash10-05vx-9100203430-9e39d5da3afdd3a15f91 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 40V, Negative-QTOF | splash10-00b9-9101401200-719cb55952a06d96d3c8 | 2021-09-22 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 10V, Positive-QTOF | splash10-03di-0000000090-02de4c2ee732e6d50b2d | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 20V, Positive-QTOF | splash10-01p9-1901002440-79ee9e61778bd08a750f | 2021-09-23 | Wishart Lab | View Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - Acetyl-CoA 40V, Positive-QTOF | splash10-0udi-0209000000-84f7f68fc2e83a8ae770 | 2021-09-23 | Wishart Lab | View Spectrum |
NMR Spectra| Spectrum Type | Description | Deposition Date | Source | View |
|---|
| Predicted 1D NMR | 1H NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 100 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 1000 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 200 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 300 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 400 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 500 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 600 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 700 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 800 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 1H NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum | | Predicted 1D NMR | 13C NMR Spectrum (1D, 900 MHz, D2O, predicted) | 2021-09-25 | Wishart Lab | View Spectrum |
| Show more...
|---|
| General References | - Blank ML, Smith ZL, Fitzgerald V, Snyder F: The CoA-independent transacylase in PAF biosynthesis: tissue distribution and molecular species selectivity. Biochim Biophys Acta. 1995 Feb 9;1254(3):295-301. [PubMed:7857969 ]
- Wysocki SJ, Wilkinson SP, Hahnel R, Wong CY, Panegyres PK: 3-Hydroxy-3-methylglutaric aciduria, combined with 3-methylglutaconic aciduria. Clin Chim Acta. 1976 Aug 2;70(3):399-406. [PubMed:947633 ]
- Al-Buheissi SZ, Patel HR, Meinl W, Hewer A, Bryan RL, Glatt H, Miller RA, Phillips DH: N-Acetyltransferase and sulfotransferase activity in human prostate: potential for carcinogen activation. Pharmacogenet Genomics. 2006 Jun;16(6):391-9. [PubMed:16708048 ]
- Michno A, Skibowska A, Raszeja-Specht A, Cwikowska J, Szutowicz A: The role of adenosine triphosphate citrate lyase in the metabolism of acetyl coenzyme a and function of blood platelets in diabetes mellitus. Metabolism. 2004 Jan;53(1):66-72. [PubMed:14681844 ]
- Griffin MJ, Sul HS: Insulin regulation of fatty acid synthase gene transcription: roles of USF and SREBP-1c. IUBMB Life. 2004 Oct;56(10):595-600. [PubMed:15814457 ]
- Putman CT, Spriet LL, Hultman E, Dyck DJ, Heigenhauser GJ: Skeletal muscle pyruvate dehydrogenase activity during acetate infusion in humans. Am J Physiol. 1995 May;268(5 Pt 1):E1007-17. [PubMed:7762627 ]
- Szutowicz A, Tomaszewicz M, Jankowska A, Madziar B, Bielarczyk H: [Mechanisms of selective vulnerability of cholinergic neurons to neurotoxic stimuli]. Postepy Hig Med Dosw. 1999;53(2):263-75. [PubMed:10355292 ]
- Ingebretsen OC, Bakken AM, Farstad M: The content of coenzyme A, acetyl-CoA and long-chain acyl-CoA in human blood platelets. Clin Chim Acta. 1982 Dec 23;126(3):307-13. [PubMed:7151284 ]
- Michno A, Raszeja-Specht A, Jankowska-Kulawy A, Pawelczyk T, Szutowicz A: Effect of L-carnitine on acetyl-CoA content and activity of blood platelets in healthy and diabetic persons. Clin Chem. 2005 Sep;51(9):1673-82. Epub 2005 Jul 14. [PubMed:16020499 ]
- Constantin-Teodosiu D, Peirce NS, Fox J, Greenhaff PL: Muscle pyruvate availability can limit the flux, but not activation, of the pyruvate dehydrogenase complex during submaximal exercise in humans. J Physiol. 2004 Dec 1;561(Pt 2):647-55. Epub 2004 Oct 7. [PubMed:15579544 ]
- Crystal HA, Davies P: Cortical substance P-like immunoreactivity in cases of Alzheimer's disease and senile dementia of the Alzheimer type. J Neurochem. 1982 Jun;38(6):1781-4. [PubMed:6176686 ]
- Evans MK, Savasi I, Heigenhauser GJ, Spriet LL: Effects of acetate infusion and hyperoxia on muscle substrate phosphorylation after onset of moderate exercise. Am J Physiol Endocrinol Metab. 2001 Dec;281(6):E1144-50. [PubMed:11701427 ]
- Peters SJ: Regulation of PDH activity and isoform expression: diet and exercise. Biochem Soc Trans. 2003 Dec;31(Pt 6):1274-80. [PubMed:14641042 ]
- Roe CR, Sweetman L, Roe DS, David F, Brunengraber H: Treatment of cardiomyopathy and rhabdomyolysis in long-chain fat oxidation disorders using an anaplerotic odd-chain triglyceride. J Clin Invest. 2002 Jul;110(2):259-69. [PubMed:12122118 ]
- Skibowska A, Raszeja-Specht A, Szutowicz A: Platelet function and acetyl-coenzyme A metabolism in type 1 diabetes mellitus. Clin Chem Lab Med. 2003 Sep;41(9):1136-43. [PubMed:14598862 ]
- Girard J: [Contribution of free fatty acids to impairment of insulin secretion and action. mechanism of beta-cell lipotoxicity]. Med Sci (Paris). 2005 Dec;21 Spec No:19-25. [PubMed:16598900 ]
- Szutowicz A, Jankowska A, Tomaszewicz M: [Disturbances of glucose metabolism in epilepsy and other neurodegenerative diseases]. Neurol Neurochir Pol. 2000;34 Suppl 8:59-66. [PubMed:11780590 ]
- Spriet LL, MacLean DA, Dyck DJ, Hultman E, Cederblad G, Graham TE: Caffeine ingestion and muscle metabolism during prolonged exercise in humans. Am J Physiol. 1992 Jun;262(6 Pt 1):E891-8. [PubMed:1616022 ]
- Constantin-Teodosiu D, Carlin JI, Cederblad G, Harris RC, Hultman E: Acetyl group accumulation and pyruvate dehydrogenase activity in human muscle during incremental exercise. Acta Physiol Scand. 1991 Dec;143(4):367-72. [PubMed:1815472 ]
- Boden G, Jadali F, White J, Liang Y, Mozzoli M, Chen X, Coleman E, Smith C: Effects of fat on insulin-stimulated carbohydrate metabolism in normal men. J Clin Invest. 1991 Sep;88(3):960-6. [PubMed:1885781 ]
| Show more...
|---|